Abstract
Letter to Editor
The Daring of Biosimilars
Nuris Ledón* and Agustín Lage
Published: 01 May, 2017 | Volume 1 - Issue 1 | Pages: 017-020
The so-called “biotechnological revolution” is changing the structure of the Pharmacopoeia [1]. The space of biological products, previously limited to blood products and vaccines, grew from the introduction of first recombinant therapeutics in the 1980s until attaining a 25% by value of the pharmaceutical market. This share is expected to reach 50% in the coming years. More than 80 biotechnology drugs have entered the market in the last ten years. It is estimated that there are more than 900 biological products on development for more than 100 diseases [2].
Read Full Article HTML DOI: 10.29328/journal.hjb.1001002 Cite this Article Read Full Article PDF
References
- Niosi J. Imitation and innovation new biologics, biosimilars and biobetters. Technology Analysis & Strategic Management. 2017; 29: 251-262. Ref.: https://goo.gl/1thHhm
- Evens R, Kaitin K. The evolution of biotechnology and its impact on health care. Health Aff. 2015; 34: 210-219. Ref.: https://goo.gl/m1Zfoi
- Richardson E. Biosimilars: To Encourage Competition, the Health Care Law Directs the FDA to Develop an Accelerated Approval Pathway for Follow-on Versions of Original Biologic Products. Health Aff. 2013; 1-5. Ref.: https://goo.gl/ZVhvQ7
- Kesselheim AS, Darrow JJ. Hatch-Waxman Turns 30: Do we need a Re-Designed Approach for the Modern Era? Yale J Health Policy Law Ethics. 2015; 15: 293-347. Ref.: https://goo.gl/m60BcS
- Walsh G. Biopharmaceutical Benchmarks. Nat Biotechnol. 2014; 32: 992-1000. Ref.: https://goo.gl/yCu0EM
- Schietl M, Stangler T, Torella C, Čepeljnik T, Toll H, et al. Acceptable Changes in Quality Attributes of Glycosilated Biopharmaceuticals. Nat Biotechnol. 2011; 29: 310-312. Ref.: https://goo.gl/R0vV5m
- EMA site. Available April 2017. Ref.: https://goo.gl/VAdNL
- Grabowski HG, Guha R, Salgado M. Regulatory and cost barriers are likely to limit biosimilar development and expected savings in the near future. Health Aff. 2014; 33: 1048-1057. Ref.: https://goo.gl/ZrNzbC
- Combest AJ, Wang S, Healey BT, Reitsma DJ. Alternative Statistical Strategies for Biosimilar Drug Development. Generic and Biosimilar Initiative Journal. 2014; 3: 13-20. Ref.: https://goo.gl/4Bs5ji
Figures:
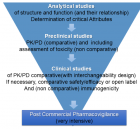
Figure 1
Similar Articles
-
Overview of Interferon: Characteristics, signaling and anti-cancer effectKangjian Zhang*,Huaiyuan Wang,Haijun Hu. Overview of Interferon: Characteristics, signaling and anti-cancer effect. . 2017 doi: 10.29328/journal.hjb.1001001; 1: 001-016
-
The Daring of BiosimilarsNuris Ledón*,Agustín Lage. The Daring of Biosimilars. . 2017 doi: 10.29328/journal.hjb.1001002; 1: 017-020
-
The Bacterial Heterotrimeric Amidotransferase GatCAB: functions, structures and mechanism-based inhibitorsVan Hau Pham*,Jacques Lapointe. The Bacterial Heterotrimeric Amidotransferase GatCAB: functions, structures and mechanism-based inhibitors. . 2017 doi: 10.29328/journal.hjb.1001003; 1: 021-032
-
Concise Review: Considerations for the Formulation, Delivery and Administration Routes of BiopharmaceuticalsAmir Mohammed Alsharabasy*. Concise Review: Considerations for the Formulation, Delivery and Administration Routes of Biopharmaceuticals. . 2017 doi: 10.29328/journal.hjb.1001004; 1: 033-053
-
Is advanced Coupling Methods best fitted in Biosensing of Microparticles?Alexander E Berezin*. Is advanced Coupling Methods best fitted in Biosensing of Microparticles?. . 2017 doi: 10.29328/journal.hjb.1001005; 1: 054-060
-
Nutritional Value of Three Different Oyster Mushrooms Grown on Cattail Weed SubstrateRam Naraian*,Bharti Dixit. Nutritional Value of Three Different Oyster Mushrooms Grown on Cattail Weed Substrate. . 2017 doi: 10.29328/journal.hjb.1001006; 1: 061-066
-
Pros and Cons Controversy on Molecular Imaging and Dynamics of Double-Standard DNA/RNA of Human Preserving Stem Cells-Binding Nano Molecules with Androgens/Anabolic Steroids (AAS) or Testosterone Derivatives through Tracking of Helium-4 Nucleus (Alpha Particle) Using Synchrotron RadiationAlireza Heidari*. Pros and Cons Controversy on Molecular Imaging and Dynamics of Double-Standard DNA/RNA of Human Preserving Stem Cells-Binding Nano Molecules with Androgens/Anabolic Steroids (AAS) or Testosterone Derivatives through Tracking of Helium-4 Nucleus (Alpha Particle) Using Synchrotron Radiation. . 2017 doi: 10.29328/journal.hjb.1001007; 1: 067-100
-
ABC and MFS Transporters: A reason for Antifungal drug resistanceNeelabh*,Karuna Singh. ABC and MFS Transporters: A reason for Antifungal drug resistance. . 2018 doi: 10.29328/journal.abb.1001009; 2: 001-007
-
Significant influence for vitamin K on different metabolic diseases according to positive effect on levels of both vitamin D, and calciumWalaa Fikry Elbossaty*. Significant influence for vitamin K on different metabolic diseases according to positive effect on levels of both vitamin D, and calcium. . 2018 doi: 10.29328/journal.abb.1001010; 2: 008-011
-
Effects of KCl (rpm/Heat) on Bacterial Protease Production in E. coli, P. aeruginosa and E. faecalisHüseyin Kahraman*,Canan Cennet Karaderi. Effects of KCl (rpm/Heat) on Bacterial Protease Production in E. coli, P. aeruginosa and E. faecalis. . 2018 doi: 10.29328/journal.abb.1001011; 2: 012-017
Recently Viewed
-
Drug Rehabilitation Centre-based Survey on Drug Dependence in District Shimla Himachal PradeshKanishka Saini,Palak Sharma,Bhawna Sharma*,Atul Kumar Dubey,Muskan Bhatnoo,Prajkta Thakur,Vanshika Chandel,Ritika Sinha. Drug Rehabilitation Centre-based Survey on Drug Dependence in District Shimla Himachal Pradesh. J Addict Ther Res. 2025: doi: 10.29328/journal.jatr.1001032; 9: 001-006
-
Feature Processing Methods: Recent Advances and Future TrendsShiying Bai,Lufeng Bai*. Feature Processing Methods: Recent Advances and Future Trends. J Clin Med Exp Images. 2025: doi: 10.29328/journal.jcmei.1001035; 9: 010-014
-
Relationship between Vitamin D Deficiency and Lipopolysaccharides Porphyromonas gingivalis Bacteria in Stunting ChildrenErwin Gunawan*,Ria Puspitawati. Relationship between Vitamin D Deficiency and Lipopolysaccharides Porphyromonas gingivalis Bacteria in Stunting Children. Ann Biomed Sci Eng. 2024: doi: 10.29328/journal.abse.1001033; 8: 059-065
-
Addiction to self-strangulation: a case-reportAurely Ameller*,Yann Le Strat,Marion Cadranel,Celine Portalier, Caroline Dubertret. Addiction to self-strangulation: a case-report . J Addict Ther Res. 2017: doi: 10.29328/journal.jatr.1001003; 1: 016-021
-
Nanoencapsulated Extracts from Leaves of Bauhinia forficata Link: In vitro Antioxidant, Toxicogenetic, and Hypoglycemic Activity Effects in Streptozotocin-induced Diabetic MiceBárbara Verônica Cardoso de Souza, Alessandra Braga Ribeiro*, Rita de Cássia Meneses Oliveira, Julianne Viana Freire Portela, Ana Amélia de Carvalho Melo Cavalcante, Esmeralda Maria Lustosa Barros, Luís Felipe Lima Matos, Tarsia Giabardo Alves, Maria. Nanoencapsulated Extracts from Leaves of Bauhinia forficata Link: In vitro Antioxidant, Toxicogenetic, and Hypoglycemic Activity Effects in Streptozotocin-induced Diabetic Mice. Arch Pharm Pharma Sci. 2024: doi: 10.29328/journal.apps.1001063; 8: 100-115
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."